The Characteristics of Low-temperature Lithium Batteries
Since entering the market, lithium batteries have been widely used for their long life and large capacity with no memory. With the expansion of batteries in various applications and fields, many problems have risen: reduced capacity, severe voltage attenuation, fast life-cycle degradation, and lithium plating just to name a few. For instance, the operating temperature of traditional lithium-ion batteries is between -20°C to 55° C. In aerospace, automotive, and special fields, these batteries must operate normally at -40° C. This becomes an issue when the discharge capacity of low-temperature lithium-ion batteries is only about 31.5% at room temperature. It is thus of great importance that we improve the low-temperature properties of low-temperature lithium batteries.
1. Factors that limit the performance of low-temperature, lithium-ion batteries
There are several factors that restrict the performance of lithium-ion batteries in low-temperature environments. The viscosity of the electrolytes in a battery increase in response to low temperatures and the electrolytes can even solidify. The compatibility between the electrolytes, negative electrodes and the separator also deteriorates. Severe precipitation occurs at the negative electrode of a lithium-ion battery. The precipitated lithium reacts with the electrolytes, and the deposition ultimately causes the SEI (Solid Electrolyte Interface) to increase in thickness. The diffusion system of a lithium-ion battery inside the active material decreases, and the Rct (Charge Transfer Resistance) increases significantly. All these factors serve to severely lower the performance of low-temperature, lithium-ion batteries.
2. The decisive factors that affect the low-temperature performance
(1)Electrolytes
The electrolyte has the greatest impact on the low-temperature performance of lithium-ion batteries. This is in large part due to the effect of the composition and physical and chemical properties of the electrolyte. As stated earlier, cycling a battery at low temperatures will cause the viscosity of the electrolytes in a battery to increase. In addition, the ion conduction speed will slow down, which ultimately causes the mismatch of the electromigration speed of the external circuit. This results in the polarization of the battery and the charge and the sharp decrease in capacity. The problem is further compounded when dendrites form on the surface of a negative electrode (this occurs easily when charging at low temperatures), which results in battery failure. The low-temperature performance of electrolytes is closely related to the conductivity of electrolytes. An electrolyte with high conductivity can transfer ions quickly and can exhibit more capacity at low temperatures. The more the lithium salt dissociates in an electrolyte, the more the number of migrations and the higher the electrical conductivity. With a higher conductivity comes a faster ion conduction rate, smaller polarization, and better battery performance at low temperatures.
Therefore, higher electrical conductivity is a necessary condition for achieving a high-performing, low-temperature lithium-ion battery. One of the ways to improve the conductivity of an electrolyte is to reduce the viscosity of the solvent. The conductivity of the electrolyte is related to the composition of the electrolyte, and reducing the viscosity of the solvent is one of the ways to improve the conductivity of the electrolyte. The good fluidity of the solvent at low temperature is the guarantee of ion transport, and the solid electrolyte membrane formed by the electrolyte at the negative electrode at low temperature is also the key to affect lithium-ion conduction. RSEI (Resistant Solid Electrolyte Interface) is the main impedance of lithium-ion batteries in low-temperature environments.
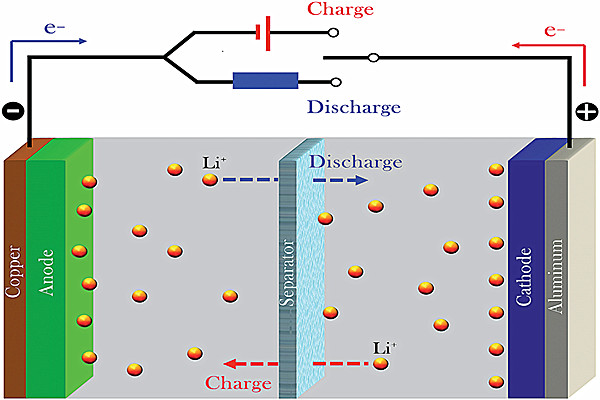
(2) Li + diffusion resistance
The other main factor limiting the low-temperature performance of lithium-ion batteries is the sharply increased Li + diffusion resistance at low temperatures, not the SEI film.
3. Low-temperature characteristics of different materials in lithium-ion batteries
Low-temperature characteristics of layered cathode materials
The layered structure of cathode materials has the unparalleled performance rate of a one-dimensional lithium-ion diffusion channel and the structural stability of a three-dimensional channel. It is the earliest commercially available lithium-ion battery cathode material. Its representative substances are LiCoO2, Li (Co1-xNix) O2, Li (Ni, Co, Mn) O2 and so on. Experts took LiCoO2 / MCMB as the research object and tested its low-temperature charge and discharge characteristics. The results show that as the temperature decreases, the discharge platform decreases from 3.762V (0 ° C) to 3.207V (-30 ° C); the total battery capacity also sharply decreases from 78.98mA · h (0 ° C) to 68.55mA · h (–30 ° C).
Low-temperature characteristics of spinel anode materials
The spinel structure LiMn2O4 cathode material has the advantages of low cost and no toxicity because it does not contain any Cobalt. However, the variability of the valency of Manganese and the Jahn-Teller effect of Mn3 + lead to issues with structural instability and poor reversibility of this component. Different preparation methods have a greater impact on the electrochemical performance of LiMn2O4 cathode materials. Take the Act as an example: the Act of LiMn2O4 synthesized by the high-temperature solid-phase method is significantly higher than that of the Sol-Gel method. This phenomenon also has a lithium-ion diffusion coefficient mainly due to the different synthetic methods that have a greater impact on the crystallinity and morphology of the product.
Low-temperature characteristics of phosphate anode materials
LiFePO4 has become the main body of the positive electrode material of batteries due to its excellent safety and stability in volume. However, the performance is not ideal due to its material as an insulator, low electronic conductivity, poor lithium-ion diffusivity, and poor conductivity at low temperatures. All these issues increase the internal resistance of the battery, which greatly affects the polarization and blocks the charge and discharge of the battery. When studying the charge and discharge behavior of a LiFePO4 battery at low temperatures, it was found that its Coulomb efficiency decreased from 100% at 55 ° C to 96% at 0 ° C and 64% at –20 ° C. The discharge voltage decreased from 3.11V at 55 ° C to -20V at 2.62V.
In other studies, nano-carbon was used to modify a LiFePO4 battery, and it was discovered that the addition of the nano-carbon as a conductive agent reduced the sensitivity of LiFePO4’s electrochemical performance, as well as low-temperature performance.
The discharge voltage of a modified LiFePO4 battery from 3.40V at 25 ° C dropped to 3.09V at –25 ° C, which is a decrease of only 9.12%. Its battery efficiency at –25 ° C came to 57.3%, which was higher than the 53.4% efficiency of a battery without nano-carbon as a conductive agent. LiMnPO4 has also recently drawn great interest. A study found that LiMnPO4 has the advantages of high potential (4.1V), no pollution, low price, and large specific capacity (170mAh / g). However, because LiMnPO4 has a lower ionic conductivity than LiFePO4, Iron is often used in practice to partially replace Manganese to form a LiMn0.8Fe0.2PO4 solid solution.
4. Low-temperature characteristics of lithium-ion battery anode materials
For the following reasons, the low-temperature deterioration of the negative electrode material of a lithium-ion battery must be taken more seriously than that of one with the positive electrode material: When charging and discharging at a low temperature and large rate, a battery undergoes severe polarization, a large deposit of lithium on the surface of the anode, and a lack of conductivity with the reaction product of lithium and the electrolyte. From the perspective of thermodynamics, the electrolyte contains a large number of polar groups such as C–O, C–N, etc., which can react with the negative electrode material, and the SEI film formed is more susceptible to low temperatures. Carbon anodes are difficult to insert lithium at low temperatures, and there is the asymmetry in the charge and discharge.
5. Research on Low-Temperature Electrolytes
The electrolyte has the role of transferring Lithium in the lithium-ion battery, and its ionic conductivity and SEI film-forming performance have a significant effect on the low-temperature performance of the battery. There are three main indicators for judging the pros and cons of low-temperature electrolytes: the ionic conductivity, electrochemical window, and electrode reactivity. The level of these three indicators depends largely on their constituent materials: solvents, electrolytes (lithium salts), and additives. Compared to the chain carbonate, the low-temperature characteristics of the EC-based electrolyte have a tighter cyclic carbonate structure, a larger force, and a higher melting point and viscosity.
However, the large polarity brought by the ring structure tends to have a large dielectric constant. EC solvents have a large dielectric constant, high ionic conductivity, and excellent film-forming properties, which effectively prevent the co-insertion of solvent molecules, making them indispensable. Therefore, most common low-temperature electrolyte systems are based on EC and the mixed Low melting point small molecule solvent. The lithium salt is an important component of the electrolyte. Lithium salts in the electrolyte not only improve the ionic conductivity of the solution but also reduce the diffusion distance of Li + in the solution. In general, the greater the Li + concentration in the solution, the greater its ionic conductivity. However, the concentration of lithium ions in the electrolyte and the concentration of lithium salts are not linearly related but are parabolic. This is because the lithium-ion concentration in the solvent depends on the dissociation of the lithium salt in the solvent and the strength of the association.
6. Process factors
In addition to the battery composition itself, the process factors in the operation of a battery have a great impact on battery performance.
(1) Preparation process
Experts studied the effect of electrode load and coating thickness on the low-temperature performance of LiNi0.6Co0.2Mn0.2O2 / Graphite batteries and found that, in terms of capacity retention, the smaller the electrode load, the thinner the coating layer and the better its low-temperature performance.
(2) Charge and discharge status
Experts have studied the impact of low-temperature charge-discharge conditions on battery cycle-life and found that larger discharge depths will cause greater capacity loss and reduce cycle life.
(3) Other factors
There is a multitude of other factors that affect the low-temperature performance of lithium-ion batteries: the surface area, pore diameter, electrode density, separator, and wettability of the electrode and electrolyte. The impact of material and process defects on the low-temperature performance of a battery cannot be ignored as well.
7. Assurance of low-temperature performance of lithium batteries
To ensure the low-temperature performance of lithium-ion batteries, the following points need to be achieved:
(1) SEI film
The formation of a thin and dense SEI film.
(2) The active material
A large diffusion coefficient of the Li + in the active material.
(3) ionic conductivity
High ionic conductivity of the electrolyte at low temperatures. Research on low-temperature batteries has also begun into another type of lithium-ion battery: The all-solid-state battery. Compared to a conventional lithium-ion battery, all-solid-state batteries, especially those with thin-film, are expected to completely solve the issue of capacity degradation and the cycle safety of batteries used at low temperatures. If you are interested in low-temperature lithium batteries, please contact the Grepow office at info@grepow.com. More information can be found at https://www.grepow.com/
Related Articles
-

Vatican Drone Show: Where Technology Meets Faith
2025-09-15 -

New Release: Tattu TA300 Multi-Channel Smart Charger for Drone Soccer
2025-09-12 -

How to Handle Battery Overheating Issues?
2025-08-12
Related products
-

-40℃ Low Temperature LiFePO4 Battery
-

-50℃ to 50℃ Low Temperature Lipo Battery
-

-40℃ to 60℃ Low Temperature Lipo Battery