Different types of Lithium Polymer batteries
Introduction
A lithium-ion polymer (LiPo) battery is a chemical battery with high energy density, lightweight, and a possibility of being made in a variety of shapes and sizes.
The lithium-polymer battery uses a file alloy as the positive electrode, a polymer conductive material, poly-acetylene, poly-aniline, or poly-p-phenol as the negative electrode, and an organic solvent as the electrolyte. The specific energy of the lithium poly-aniline battery can reach 350Wh / kg, but the specific power is only 50-60W / kg, the use temperature is -40-70℃, and the cycle life is about 330 cycles. LiPo batteries can also be made ultra-thin and customized to different shapes and capacities to meet the needs of the products they power. Grepow’s shaped batteries can have a minimum thickness of 0.4mm in many special shapes.
Types of lithium-ion polymer batteries
When we talk about different types of LiPo batteries, we are referring to batteries that modify their positive electrode materials to achieve different energy densities, capacities, and temperature resistance; depending on the material, they can be used in different applications. These are also known as different chemistries. The four main chemistries for LiPo batteries are the following:
* Lithium cobalt oxide battery (LiCoO2, LCO battery)
* Lithium-ion ternary battery (NCA, NMC battery)
* Lithium-ion manganese oxide battery battery(LiMn2O4, LMO battery)
* Lithium iron phosphate battery (LiFePO4, LFP battery)
Lithium cobalt oxide battery (LiCoO2, LCO battery)

Lithium cobalt oxide is the earliest commercialized lithium battery cathode material. The lithium cobalt oxide battery has a stable structure, high capacity ratio, and outstanding comprehensive performance. The nominal voltage is 3.7V.
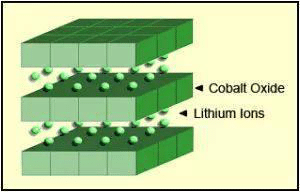
LiCoO2 is used as the positive electrode material. The theoretical capacity is 274mAh / g, the actual capacity is about 140mAh / g, and there are reports that the actual capacity has reached 155mAh / g. The main advantages of the positive electrode material are the following: higher working voltage (average working voltage is 3.7V), stable charge and discharge voltage, suitable for large current charge and discharge, high specific energy, good cycle performance, high conductivity, simple production process and easy preparation.
Spider chart of LCO battery
The disadvantages are the following: It has a high price, poor overcharge resistance, and poor safety, and the cycle performance needs to be further improved. Compared with other lithium polymer battery systems, lithium cobalt oxide has the disadvantages of having a relatively short life, low thermal stability, and limited load capacity (specific power). Like other cobalt hybrid lithium-ion batteries, lithium cobaltate uses graphite anodes. Their cycle life is mainly limited by the Solid Electrolyte Interface (SEI), which generally manifests in the gradual thickening of the SEI film and plating from rapid or low-temperature charging.
Therefore, in order to increase the life of the battery, nickel, manganese and/or aluminum have been added to increase the load capacity and reduce the cost. This is the ternary lithium battery.
Lithium-ion ternary battery (NCA, NMC battery)
The ternary polymer lithium battery consists of ternary cathode material of nickel cobalt manganate (Li (NiMnCo) O2) or nickel cobalt lithium aluminate (NCA). The ternary composite cathode material is nickel (Ni), cobalt (Co), manganese (Mn) or aluminum (Al); the proportion of nickel, cobalt, and manganese can be adjusted according to actual needs, and different capacity and magnification targets can be achieved, which includes (but is not limited to):
* 333 system (the ratio of nickel, cobalt, and manganese is 1: 1: 1)
* 523 system (the ratio of nickel, cobalt, and manganese is 5: 2: 3)
* 622 system
* 811 system
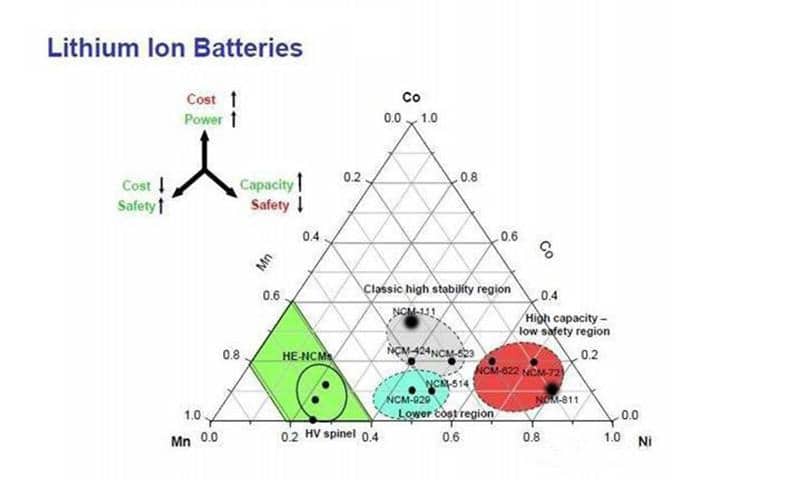
Compared to the lithium cobalt oxide battery, a ternary lithium battery is safe, but the voltage is too low, and the nominal voltage is only 3.6V. When used in mobile phones (the cut-off voltage of mobile phones is generally around 3.0V), there is obvious capacity shortage. The ternary materials are mainly nickel-cobalt lithium aluminate (NCA) and nickel-cobalt manganese lithium (NMC); NCA has the highest specific capacity among the commercial cathode materials.
NCA (Lithium Nickel-Cobalt Aluminate) battery
The NCA battery has been used since 1999. It has higher specific energy, quite good specific power and a long service life similar to NMC. High energy, power density and good cycle life also make the NCA battery a candidate for electric vehicles. Its safety and cost is less flattering.
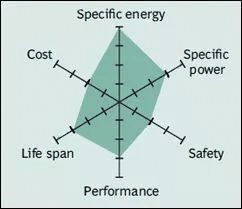
Spider chart of NCA battery
NMC (Lithium Nickel-Cobalt Manganese) battery
The NMC battery has good overall performance and performs well in terms of specific energy. The combination of nickel and manganese are significant: nickel is known for its high specific energy, but its stability is poor; the manganese spinel structure can achieve low internal resistance but has low specific energy. For this reason, these two active metals have complementary advantages. This battery is the main power for electric vehicles, and Tesla uses it especially as it has the lowest self-heating rate. Similarly, NMC is the battery of choice for power tools, electric bicycles and other electrical power systems. The cathode combination is usually one-third nickel, one-third manganese and one-third cobalt, also known as the 333 system (1: 1: 1). This provides a unique blend, which also reduces raw material costs due to the reduced cobalt content. Another successful combination is the 532 system, which contains 5 parts nickel, 3 parts cobalt and 2 parts manganese (5: 3: 2). Due to the high cost of cobalt, battery manufacturers have shifted from cobalt to nickel cathodes. Nickel-based systems have higher energy density, lower cost, and longer cycle life than cobalt-based batteries, but their voltage is slightly lower.
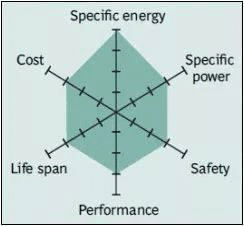
Spider chart of NMC battery
Lithium-ion manganese oxide (LiMn2O4, LMO) battery

The production process of lithium manganese oxide batteries is mature, and it is easier to carry the 18650 type batteries used in Japan and South Korea. LiMn2O4 is used for the cathode material of these lithium-ion batteries. The theoretical capacity is 148mAh / g, and the actual capacity is 90 to 120mAh / g. The operating voltage range is 3 to 4V. The main advantages of the positive electrode material are its rich manganese resources, low price, high safety, and relatively easy preparation. LMO batteries can be discharged at a current of 20-30A and have moderate heat accumulation. Up to 50A 1 second load pulse can also be applied. Continuous high load at this current will cause heat to accumulate, so the battery temperature must not exceed 80℃ (176℉). It is mostly used in power tools, medical equipment, and hybrid and pure electric vehicles.
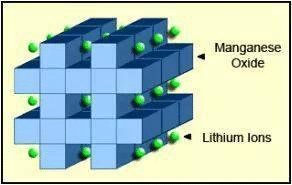
The disadvantages of the lithium manganese battery is that the theoretical capacity is relatively lower, and the capacity is about one third lower than that of lithium cobalt oxide. The material will slowly dissolve in the electrolyte, reflecting how the compatibility with the electrolyte is not very good. During deep charging and discharging, the material is prone to lattice change, causing the battery capacity to decay quickly especially when used at higher temperatures.
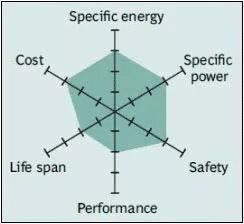
Spider chart of LMO battery
The characteristic parameters of LMO batteries are not ideal, but the new design has improved in terms of power, safety and life. Therefore, pure LMO batteries are no longer common today; they are only used in special cases.
Lithium iron phosphate (LiFePO4, LFP) battery
Lithium iron phosphate has good electro-chemical performance and low resistance. This is achieved through nano-scale phosphate cathode materials. The main advantages are high rated current, long cycle life, good thermal stability, enhanced safety and tolerance for excessive use.It also has good safety, long cycle life, abundant raw material resources and less environmental pollution. But the shortcomings are also obvious. In addition to the extremely poor cycling performance at low temperatures (except the special formula for high and low-temperature resistance), it has low conductivity and tap density, and its energy density is only 120-150wh / kg. Lithium phosphate is commonly used instead of lead-acid starting batteries. Four batteries in a series produce 12.80V (4S), which is similar to the voltage of six 2V lead-acid batteries connected in series. A vehicle charges lead-acid to 14.40V (2.40V / battery) and maintains the float charge state (the purpose of float charging is to maintain a full charge level and prevent sulfation of lead-acid batteries). By connecting four lithium phosphate batteries in series, the voltage of each battery is 3.60V, which is the full charge voltage. At this point, the charging should be disconnected, but continue to charge while driving. Lithium phosphate tolerates some overcharging; however, since most vehicles maintain a voltage of 14.40V for long periods of time on long journeys, the mechanical stress of lithium phosphate batteries may increase. Time will tell us how long lithium phosphate can withstand overcharging as an alternative to lead-acid batteries. The low temperature will also reduce the performance of lithium ions, which may affect the starting ability in extreme cases.
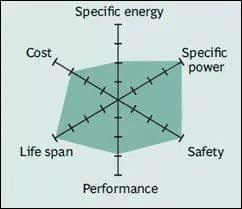
Spider chart of LiFePO4 battery
(Image source: Battery University)
Grepow’ Lithium Polymer batteries
The lithium-ion polymer batteries from Grepow have these different types of cathode materials, and they can be adjusted according to customers’ needs. Contact us at info@grepow.com if you would like a suitable recommendation or consultation for your product.
Learn more about batteries
Keep an eye out on Grepow’s official blog, where we regularly update industry-related articles to keep you up-to-date.
Grepow website: https://www.grepow.com/
Grepow Blog: https://www.grepow.com/blog.html
Related Articles
-

Building an FPV Drone: A Deep Dive into the Technology
2025-06-30 -

What is an 8S LiPo Battery?
2025-06-20 -

What Is the Voltage of a LiPo Battery?
2025-06-17
Related products
-

37000mAh Semi-Solid State High Energy Density Battery
-

50C High Discharge Battery - High C Rate LiPo
-

High C Rate LiFePO4 Battery