The Research Progress of Flexible Metal-air Battery
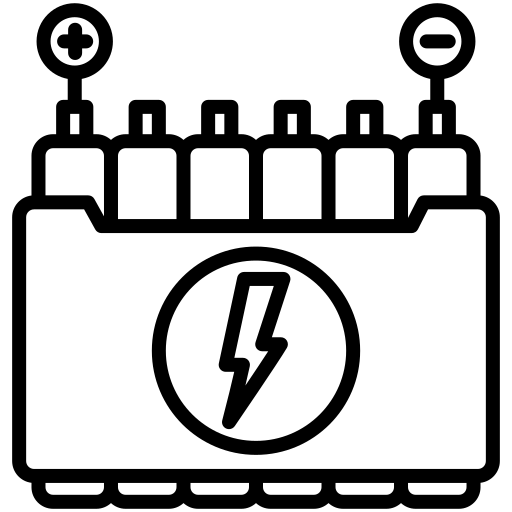
With the development of flexible and wearable electronic devices, flexible energy storage devices have attracted extensive attention from the scientific and industrial communities. In recent years, a series of works have been carried out around flexible lithium-ion batteries and supercapacitors, and important progress has been made. However, in order to extend the life of electronic devices, energy storage systems need to have a higher energy density. Flexible metal-air batteries that can be cyclically charged and discharged will greatly improve the endurance of flexible and wearable electronic devices, but more challenges need to be addressed. On the one hand, the energy density, energy efficiency, and cycle life of the battery need to be improved; on the other hand, the electrode structure, electrolyte materials, and battery structure need to be optimized to maintain stable electrochemical performance under deformation conditions. This article will introduce the recent advances in the structural design, electrode and electrolyte material development, and operating condition management of flexible metal-air batteries, mainly zinc-air and lithium-air batteries, and discusses future research directions and prospects. Metal-air batteries have attracted extensive research attention due to their high theoretical capacity and energy density as shown in Figure 1. Among them, rechargeable zinc-air batteries in alkaline systems and lithium-air batteries in organic systems as typical representatives of aqueous and non-aqueous systems are the hotspots of recent research. The working principle of the battery is shown in Figure 2. When the battery is prepared to be flexible, it is necessary to design new flexible structures, prepare flexible electrode materials, and solid electrolyte membranes to face more challenges.
Fig. 1 Comparison of capacity, energy density, and voltage of different metal-air batteries (pic: from Boris Shvartsev)
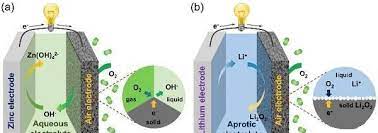
Schematic diagram of the working principle of zinc-air battery in an alkaline system and lithium-air battery in a non-aqueous system.
Flexible Battery Structure and Testing
The current cell structure widely used in flexible zinc-air and lithium-air batteries is a sandwich structure consisting of a flexible positive electrode, an electrolyte membrane, and a negative electrode stacked on top of each other. Another structure is a tubular one that uses a wire-shaped metal electrode with an electrolyte layer and an air electrode layer wrapped around the surface in turn. In addition, there are some new structures, such as a foldable battery structure and a flexible, ultra-light lithium-air battery inspired by bamboo sticks. In addition to the charge/discharge and cycle life tests in conventional batteries, stability under external forces is of paramount importance for testing flexible batteries. These include electrochemical stability under bending, twisting at different angles and stretching at different lengths, as well as retention of performance under long-term fatigue.
Metal Electrode
In flexible air batteries, metal sheets are often used directly as electrodes. However, the metal sheet may suffer from fatigue failure in long-term bending. In flexible zinc-air batteries, there is a combination of metal powder and binder and conductive carbon powder to form a composite electrode, which improves the flexibility and stability of the electrode. In flexible lithium-empty batteries, lithium metal and stainless steel mesh are rolled together to improve the fatigue resistance of the metal electrode. In addition, in order to achieve a certain stretchability in a flexible battery, the metal electrodes can be made into a spring-like material or a combination of small pieces and a complete electrode to meet the need for stretching through the "integer into zero".
Electrolyte Film
In flexible zinc-air batteries, anion exchange membranes and alkaline gel electrolytes are mainly used as the electrolyte membranes of the batteries. In flexible lithium-air batteries, the electrolyte membranes mainly include gel, solid and composite polymer electrolyte membranes. In order to achieve the good electrochemical performance of flexible batteries, the electrolyte membrane is required to have good conductivity, chemical, and electrochemical stability and other properties of traditional liquid electrolytes, in addition to the interfacial problem with metal and air electrodes is a challenge that needs to be solved. For the electrolyte membrane-metal electrode interface, the problems of dendrite and surface passivation need to be overcome. For the electrolyte membrane-air electrode interface, the solid electrolyte greatly reduces the effective reaction interface. For lithium-air batteries, the degradation of the reaction area is further exacerbated by the fact that the product is solid lithium peroxide. Thus, effective methods are needed to increase the reaction interface. In addition, during the bending or twisting of the battery, the electrode and electrolyte membrane may separate due to differences in the mechanical properties of the electrode and electrolyte membranes. How to maintain the stability of the interface is the key to ensure the long-term stable operation of the battery.
Air Electrode
Air electrodes, as an important component of metal-air batteries, have been the focus of research. On the one hand, an effective catalyst is needed to achieve rapid charging and discharging of the battery; on the other hand, a suitable structure is needed to ensure oxygen transport. In flexible batteries, it is even more necessary for the electrode to have good flexibility to meet the needs of deformation. Currently, the main flexible electrodes include:
Electrodes consisting of a network of carbon cloth or carbon fiber.
e.g. carbon nanotube paper, graphite paper electrodes made of carbon nanotube materials (e.g. carbon nanotubes, graphene).
Electrodes formed from metal substrates such as stainless steel mesh, nickel mesh.
Some other new flexible electrodes.
Operation Management
Typically, zinc-air batteries operate directly in the air, while lithium-air batteries operate in oxygen. The operating conditions can seriously affect the performance of the battery. First of all, moisture in the air will affect the stability of the electrolyte film, while carbon dioxide in the air has a greater impact: carbonate will be formed in zinc-air batteries, affecting the conductivity of the electrolyte; and lithium carbonate will be formed as a solid by-product in lithium-air batteries, affecting the charging performance of the battery. Secondly, battery performance is usually tested at room temperature, while the actual use of the temperature has a large variation. For example, in wearable devices, the operating temperature of the battery may rise to thirty degrees or more due to contact with the human body. In different seasons and regions, the temperature will vary even more. Therefore, future battery testing will need to look in more detail at stability under different gas atmospheres and temperatures, and adopt appropriate management measures.
Future Developments of Flexible Batteries
In recent years, a number of advances have been made in flexible metal-air batteries, with substantial hints of the energy density, efficiency, and cycle life of the batteries. Future research needs to further address the following issues.
1.New structural design of the battery, which meets the requirement of maintaining stable electrochemical performance under various deformation conditions.
2.The establishment of evaluation criteria to standardize the assessment of battery performance, such as the specification of accepted flexibility test standards (e.g., bending and twisting angles, tensile lengths, fatigue tests, etc.) based on uniform mass or volume.
3.development of flexible components, including metal and air electrodes, electrolyte membranes, collectors, and encapsulation materials.
4.the management of the operating conditions will ensure that stable electrochemical performance is provided under different conditions. In conclusion, future research needs to use a combination of experimental online monitoring and numerical simulation and other technical means to clearly elucidate the relationship between material transport, structural changes, and electrochemical reactions during battery operation to provide important guidance for rational battery design.
More about flexible batteries can be found on the page: https://www.grepow.com/shaped-battery.html
Contact us at info@grepow.com
Related Articles
-

Building an FPV Drone: A Deep Dive into the Technology
2025-06-30 -

What is an 8S LiPo Battery?
2025-06-20 -

What Is the Voltage of a LiPo Battery?
2025-06-17