How is the remaining 1% of your phone’s battery so durable?
Have you ever been outside when your phone’s battery ominously turned red and drained down to 1%? Without a charger, you might have panicked at the prospects of your phone turning off. You might have also found yourself surprised at how long your phone stayed on until you were able to find a charger.
In this article, we will explore how your phone’s battery stays so durable in the last 1% of its remaining capacity.
Does the voltage indicate the actual State of Charge?
In order to understand the durability of the remaining 1% of a battery, we must first explore how a battery operates. Most consumers generally believe that a low voltage means that the battery’s life is running out. At 4.2V per cell, people may assume that the lithium battery is fully charged. At 3.6V, they may think that half of the battery’s life remains, and, at 3.0V, they may think that the battery is worn out. However, this is not the complete picture of a lithium battery. Let us take a look at the graph below:
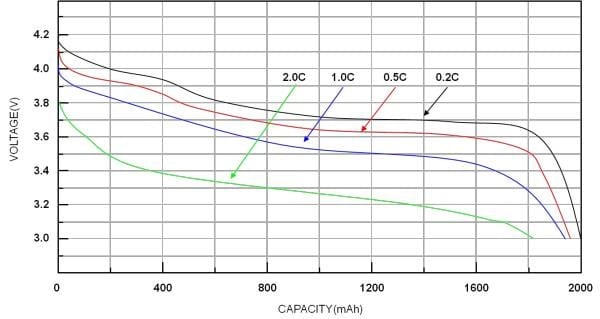
Source: “Designing Applications with Li-ion Batteries,” Richtek
This graph represents the discharge curve of a lithium-ion battery. If you follow the red line for 0.5C, you will notice that the battery has been discharged at 1500mAh at 3.6V. The remaining SOC (State of Charge) comes to 25% and not 50% as might be assumed.
Ultimately, this shows us that the discharge rate affects the remaining capacity, so the voltage does not in fact represent the State of Charge. Furthermore, the different-colored curves in the graph correspond to how much current is discharged. You will notice that with the larger discharge current comes a faster voltage drop.
However, this graph only reflects a constant current discharge and does not show how the discharge currents can vary greatly under different working conditions. Take, for example, our mobile phones’ many working states: standby, talking, Instagram, playing games, etc. Moreover, the battery will depreciate after it has been used for some time. The internal resistance will also increase, which affects the discharge curve. A battery’s capacity can also decrease as it ages. The extent to which a battery will age largely depends on its chemistry and materials (e.g. Diaphragm, Electrolyte). Its life cycles and capacity are affected by the charge and discharge rate, humidity, and temperature as well. One example is the “self-discharge” phenomenon of batteries: Even if no one is using it, it will slowly discharge itself. Another example is with the temperature’s effect on a battery.
Low temperatures in particular cause a reduced amount of electricity that can be released. It is for this reason that electrical cars break down easily in the winter and phones die more quickly in the cold. Ultimately, portable electronic devices are around us all the time, and they use lithium-based batteries. Despite how Lithium is deeply embedded in modern-day technology, the element is still a major mystery to the majority of the world. The general populace does not realize that the main rules behind its operation in batteries:
They cannot be over discharged
They cannot be overcharged
They cannot perform well in high or low-temperature environments
Features of BMS
The BMS (Battery Management System) monitors a battery’s status and protects it from short circuiting, over charging or discharging, and much more. Broken down, the BMS’s many safety and managing functions can be divided into these 4 types:
1. Communication Function
Communication Protocol(SMBus, CAN Bus)
Communication Protection
SOC Indicator
Current Detection
Abnormal Log
SelfInspection
Activity Log
2. Charge Management
3. Discharge Management
4. Others See here for a detailed account of the functions of the BMS.
Source
“Designing Applications with Li-ion Batteries” Richtek , 2014, https://www.richtek.com/battery-management/en/designing-liion.html
Learn more about batteries
Keep an eye out on Grepow’s official blog, where we regularly update industry-related articles to keep you up-to-date on the battery industry.
Grepow website: https://www.grepow.com/
Grepow Blog: https://www.grepow.com/blog.html
Related Articles
-

Practical Guide to 6S LiPo Batteries for Drones & RC Models
2025-04-23 -

How to Choose a LiPo Battery Charger?
2025-04-08 -

Join Grepow at AAA 2025+HearTECH Expo
2025-03-17