What are the Advantages of Lithium Polymer Battery?
Compared to secondary batteries such as Lead-acid batteries, Ni-Cd batteries, Ni-MH batteries, and rechargeable alkaline batteries, what are the advantages of lithium polymer battery?
The working principle of lithium polymer battery
A lithium polymer battery is actually a lithium-ion concentration difference battery. The positive and negative electrodes are a secondary battery system composed of two different substances that can be reversibly embedded and released lithium-ions. The charge and discharge of the battery are completed by the transfer of lithium ions between the positive and negative electrodes. In the charging process, Li+ is separated from the positive compound and embedded into the lattice of the negative electrode by an electrolyte. The positive electrode is in a high-potential lithium poor state, and the negative electrode is in a low-potential lithium rich state. At the same time, the compensating charge of the electron is supplied from the external circuit to the negative electrode to ensure the charge balance of the negative electrode. On the contrary, when discharging, Li+ emerges from the negative electrode, passes through the electrolyte and is embedded into the positive electrode, and the positive electrode is in the lithium-rich state. Before the naming of "lithium-ion battery", some people called the battery "rocking chair battery", because lithium-ions migrated back and forth between the positive and negative electrodes during charging and discharging. The operating voltage of a lithium polymer battery is related to the lithium-ion imbedded compound and lithium-ion concentration constituting the electrode.
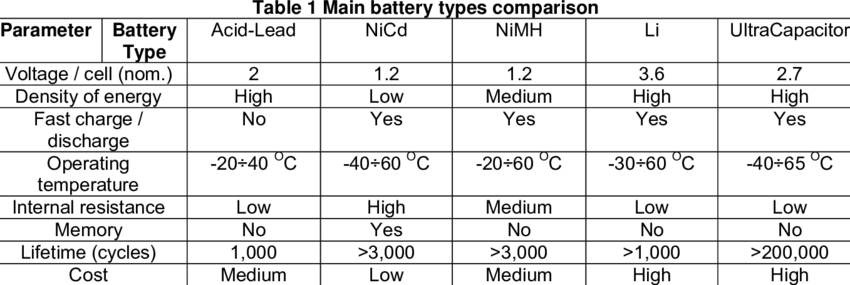
Comparison of batteries of different types
In the normal charging and discharging conditions, lithium-ions are embedded and exfoliated between the layered carbon material and the layered oxide structure, generally causing only the change of layer spacing and not damaging the crystal structure. And in the process of charging and discharging, the chemical structure of the anode material is basically unchanged. Therefore, lithium polymer battery reaction is an ideal reversible reaction from the reversibility of charge and discharge reaction. Charge and discharge principle of lithium polymer battery (taking EC/DEC solution with graphite as a negative electrode, LiCoO2 as a positive electrode and 1 mol/L LiPF6 as an electrolyte as an example)
Classification of lithium-ion batteries
There are many types of lithium-ion batteries. According to the temperature, it can be divided into a high-temperature lithium-ion battery and a normal temperature lithium-ion battery.
According to the state of the electrolyte used, it can be divided into liquid lithium-ion battery (commonly known as the lithium-ion battery), polymer electrolyte lithium-ion battery and all solid lithium-ion battery; according to the shape, can be divided into button lithium-ion battery, square lithium-ion battery, cylindrical lithium-ion battery, etc.
In the academic world, it can also be classified according to the different types of anode materials. The anode materials used include lithium cobalt oxide, lithium manganese oxide, and lithium ferrous phosphate and so on.
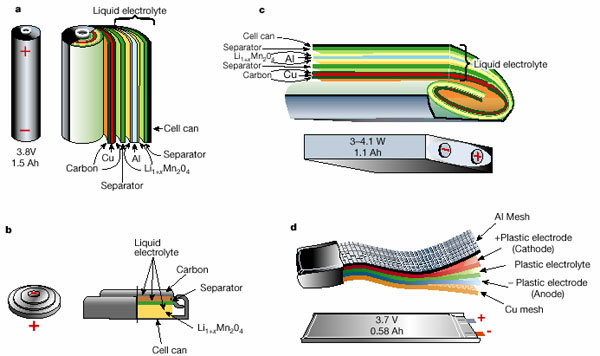
Schematic drawing showing the shape and components of various lithium-ion batteries.
a: cylinder, b: coin, c: rectangular, d: planar (lithium polymer batteries)
Characteristics
Schematic drawing showing the shape and components of various lithium-ion batteries. a: cylinder, b: coin, c: rectangular, d: planar (lithium polymer batteries) Compared to secondary batteries such as Lead-acid batteries, Ni-Cd batteries, Ni-MH batteries, and rechargeable alkaline batteries, lithium polymer battery have the following advantages:
High OCV
Currently commercialized single lithium polymer battery’s voltage platform up to 3.6-3.7V, it is equivalent to 3 times the voltage of Ni-Cd or Ni-MH battery (1.2 V). When high voltage is required, lithium-ion batteries do not only have the small series number and high space utilization but also with small mutual influence between batteries.
High energy density
At present, the actual specific energy of lithium polymer batteries has reached 140 Wh/ kg, volumetric specific energy around 300Wh/L. However, the common used Ni-Cd battery’s specific energy and volumetric specific energy is 40 Wh/kg and 125 Wh/L; Ni-MH battery’s specific energy and volumetric specific energy is 60Wh/kg and 165 Wh/L. Figure 1-1 shows the energy density comparison of various batteries.
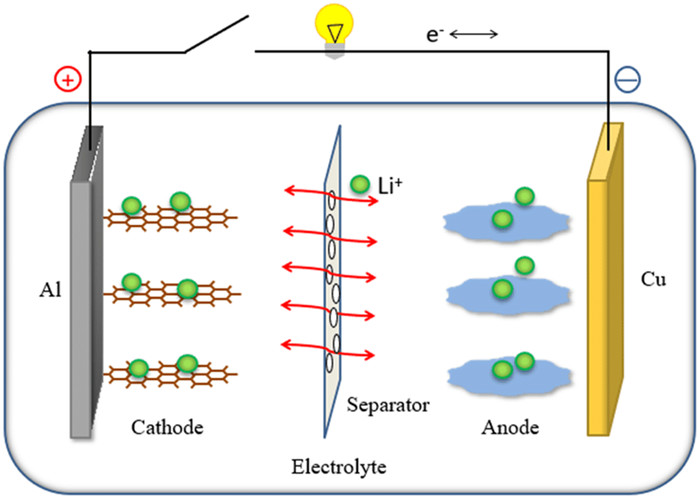
The schematic drawing of working-mechanism of lithium-ion batteries.
Greatly improved safety performance and long cycle life
lithium polymer battery uses carbon material as the negative electrode to avoid the formation of lithium dendrites during discharge, and the battery safety performance is obviously improved.
The self-discharge rate is small and no memory effect
The monthly self-discharge rate of lithium polymer battery is only 6-8%, less than half of that of Ni-Cd and Ni-MH batteries.
Environmentally friendly
Compared with traditional batteries, such as VRLA and Ni-Cd battery, which has contained toxic heavy metals, lithium polymer batteries are basically no pollution and belong to clean energy.
Fast charge and discharge
1 C charge and discharge capacity up to 80% of the capacity of 0.2 C discharge;
High-efficiency charge and discharge
After the first cycle, the Coulomb efficiency remained essentially at 100%;
Wide working temperature range
The working temperature is approximate -25 ℃~+45 ℃, with the improvement of the electrolyte and cathode material, which is expected to widen to -40 ℃~+70 ℃.
Long service life
80% DOD (depth of discharge) can be charged and discharged up to 1200 times. When using deep charge and discharge, the number of cycles can reach more than 5,000 times. If you are interested in our products, please don't hesitate to contact us at any time! Email: info@grepow.com Grepow Website: https://www.grepow.com/
Related Articles
-
Solid-State Battery VS Lipo Battery: What’s the Difference?
2024-04-12 -

How to Choose an Aerial Photography Drone?
2024-03-22 -

What Is An eVTOL And How To Choose Batteries For It?
2024-03-07
Related products
-

-50℃ to 50℃ Low Temperature Lipo Battery
-

45C High Discharge Battery - High C Rate LiPo
-

Round Lipo Battery