Basic concepts of lithium-ion polymer batteries
An Introduction to Lithium-ion batteries
A lithium-ion polymer (LiPo) battery is a family of rechargeable battery types in which lithium ions move from the negative electrode to the positive electrode during discharge and back when charging. We will explain some basic concepts for these batteries in this article.
State-Of-Charge (SOC)
The state of charge (SOC) can be defined as the state of the electrical energy available in the battery, usually expressed as a percentage. Because the available electrical energy will be different due to charge and discharge current, operating temperature and aging phenomena, the definition of SOC is also divided into two types:
Absolute State-Of-Charge (ASOC)
The ASOC is a calculated reference value based on the designed fixed capacity value when the battery is manufactured. The ASOC of a fully charged brand new battery is 100% while an aging battery cannot reach 100% even in different charging and discharging conditions while fully charged.
Relative State-Of-Charge (RSOC)
The RSOC is at 100% when the battery is fully charged and 0% when fully discharged. The figure (Figure 1) below shows the relationship between voltage and battery capacity at different discharge rates. The higher the discharge rate, the lower the battery capacity. When the temperature is low, the battery capacity will also decrease.
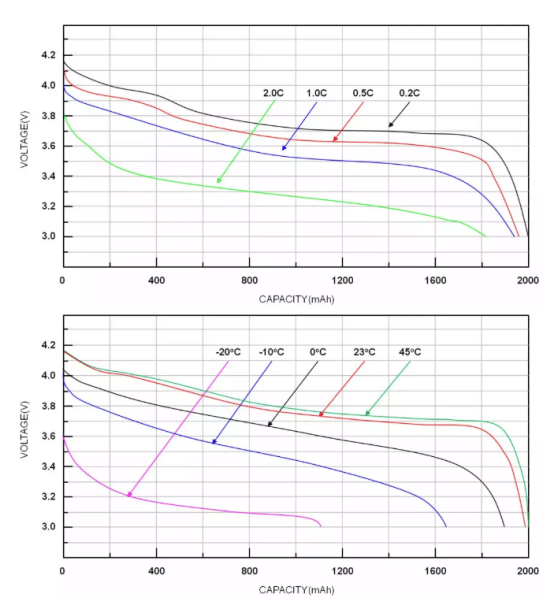
Figure 1. The relationship between voltage and capacity at different discharge rates and temperatures
Maximum Charging Voltage
The maximum charging voltage is related to the chemical composition and characteristics of the battery. The full charging voltage of a normal lithium battery is 4.2V. There are high voltage LiPo batteries with maximum charging voltages of 4.35V; there are a series of batteries from Grepow that can reach 4.45V for its maximum.
Fully Charged
When the difference between the battery voltage and the highest charging voltage is less than 100mV and the charging current is reduced to C/10, the battery can be regarded as fully charged. The battery characteristics are different, and the fully charged conditions are also different. The figure (Figure 2) below shows a typical lithium battery charging characteristic curve:
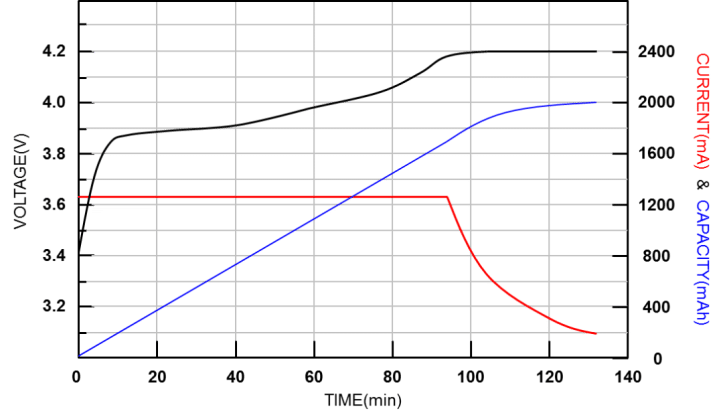
Figure 2. Charging characteristic curve of lithium battery
Minimum Discharging Voltage
The minimum discharge voltage can be defined by the cut-off discharge voltage, which is usually the voltage when SOC is 0%. This voltage value is not a fixed number and thus changes with load, temperature, aging degree or others.
Fully Discharged
When the battery voltage is less than or equal to the minimum discharge voltage, it is fully discharged.
Discharge rate (C-Rate)
The charge and discharge rate is a representation of charge-discharge current relative to the battery capacity; this is also called the C-Rate (C). If you use 1C to discharge for an hour, ideally, the battery will be completely discharged. Different charge and discharge rates will result in different available capacities. Generally, the larger the charge and discharge rate, the lower the available capacity.
Life Cycles
The life cycles is the number of times that a battery has undergone a complete charge and discharge, which can be estimated from the actual discharge capacity and designed capacity. Whenever the accumulated discharge capacity is equal to the designed capacity, the number of cycles equals one. Usually, after 500 charge and discharge cycles, the capacity of a fully charged battery drops by 10% to 20%. In general, the number of life cycles only calculates when the battery's fully charged capacity is no less than 80% of the nominal capacity.
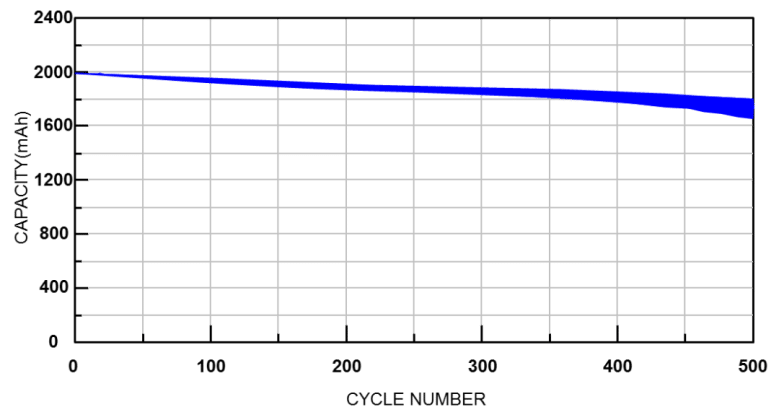
Figure 3. Relationship between the number of cycles and battery capacity
Self-Discharge
The self-discharge of all batteries will increase as the temperature increases. It is generally not a manufacturing defect but a characteristic of the battery itself. Typically, every 10℃ increase in battery temperature doubles the self-discharge rate. Lithium-ion polymer batteries have a self-discharge capacity of approximately 1 to 2% per month, while nickel-based batteries in various types have a self-discharge capacity of 10 to 15% per month.
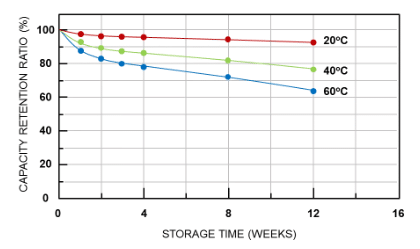
Figure 4. Performance of lithium battery self-discharge rate at different temperatures (20℃, 40℃, 60℃)
Learn more about batteries
Keep an eye out on Grepow’s official blog, where we regularly update industry-related articles to keep you up-to-date.
Grepow website: https://www.grepow.com/
Grepow Blog: https://www.grepow.com/blog.html
Related Articles
-
Solid-State Battery VS Lipo Battery: What’s the Difference?
2024-04-12 -

How to Choose an Aerial Photography Drone?
2024-03-22 -

What Is An eVTOL And How To Choose Batteries For It?
2024-03-07
Related products
-

Gens Ace Redline Series 5100mAh 7.6V 130C Hardcase RC Car LiPo Battery Pack
-

Tattu R-Line Version 5.0 1400mAh 22.2V 150C Lipo Battery Pack
-

Curved Battery